Background: Until late 2019, few treatments had been approved by the FDA for treating sickle cell disease (SCD). Voxelotor (Oxbryta®) is a sickle hemoglobin-polymerization inhibitor approved by the FDA in November 2019 for treatment of SCD in adults and adolescents aged ≥12 years under an accelerated approval based on results of the pivotal HOPE study. In HOPE, voxelotor increased average hemoglobin (Hb) by 1.1 g/dL from baseline in patients with 1-10 vaso-occlusive crises (VOCs) in the previous year and a Hb level between 5.5 and 10.5 g/dL who were not transfusion dependent. Of the participants on voxelotor 1500 mg, 51% had a Hb response >1.0 g/dL at week 24 (Vichinsky et al, NEJM 2019). This study sought to assess the real-world effectiveness of voxelotor based on data during the first 6 months post FDA approval.
Methods: Data on medical and pharmacy claims for patients who were aged ≥12 years and receiving voxelotor from December 2019 to May 2020 were obtained from the Symphony Health claims dataset. For each patient, the date of the first voxelotor claim was defined as the "index date." Patients with at least 1 year's data prior to the index date were included in the analyses. Patients' demographic and clinical characteristics were summarized descriptively. Standardized annualized rates of transfusions and VOCs per patient per year (PPPY) prior to and after voxelotor initiation were compared. A subset of patients in the Symphony Health claims dataset had Hb lab data available. Patients with at least 1 Hb result within 30 days prior to the index date and at least 1 Hb result after the index date were included for Hb analyses. For these patients, change in Hb and the percentage of patients achieving a >1 g/dL increase in Hb were summarized. Confidence intervals and P values for changes in outcomes were based on bootstrapping.
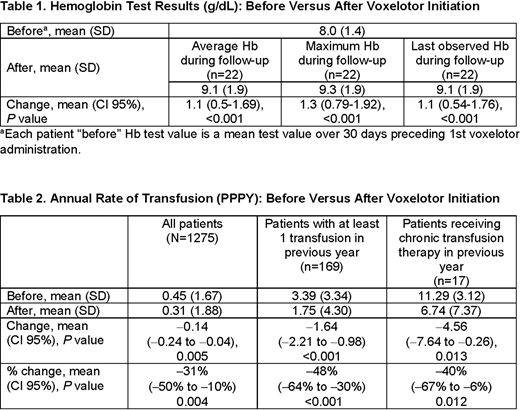
Results: As of May 31, 2020, 1275 patients from the Symphony Health claims datasets were identified who received voxelotor (40% male, mean age 35.7 years). In the year prior to voxelotor initiation, 715 (56.1%) of these patients received hydroxyurea, 121 (9.5%) received L-glutamine, 166 (13.0%) received at least 1 transfusion, 17 (1.3%) were on chronic transfusion (≥8 transfusions per year), and 681 (53.4%) had 1 or more VOCs. Mean (SD) follow-up was 64.9 (40.7) days. Among 1275 patients, 175 and 52 patients had at least 1 Hb level measurement during the 1 year prior to and after voxelotor initiation, respectively. Among the subset of patients with their Hb level tested within 30 days prior to the index date and at least 1 Hb level after index date (n=22), the baseline average Hb level was 8.0 g/dL (SD 1.4, median 7.9 g/dL, range 5.0-11.8 g/dL). Mean increase in Hb from baseline was 1.1-1.3 g/dL (Table 1) depending on the approach used to calculate Hb levels after voxelotor initiation; 55% (95% CI 32%-77%) of patients achieved a Hb increase >1 g/dL after voxelotor initiation. Among all 1275 patients, mean (SD) overall transfusion rates declined from 0.45 (1.67) PPPY pre-index to 0.31 (1.88) post-index, a change of -0.14 PPPY (P=0.005). Among 169 patients who received at least 1 transfusion in the year prior to initiation of voxelotor, the transfusion rate dropped from 3.39 (3.34) to 1.75 (4.30) PPPY, a change of -1.64 PPPY (P<0.001). Among 17 patients receiving chronic transfusions, the transfusion rate dropped from 11.29 (3.12) to 6.74 (7.37) PPPY, a change of -4.56 PPPY (P=0.013) (Table 2). After voxelotor initiation, the annualized rates of VOC were numerically reduced from 3.86 (6.69) to 3.64 (8.54) (P=0.248). To address the potential bias from the relatively short follow-up duration, similar results of transfusion and VOC rates were observed among patients with at least 30 days of follow-up or when only events within 3 months prior to index date were considered.
Conclusions: Based on the first 6 months' data after the approval of voxelotor in the US, in real-world practice, voxelotor increases Hb by at least 1 g/dL, consistent with the HOPE randomized controlled trial results. Evidence suggests that transfusion rates decreased after voxelotor initiation. A favorable downward trend in VOC rates was also observed. This real-world evidence provides additional support for the use of this novel therapy in the treatment of hemolytic anemia and its associated complications in the SCD population. Further evaluation with a larger sample size and longer follow-up will help to confirm these findings.
Zaidi:Global Blood Therapeutics: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Consultancy, Honoraria; Cyclerion: Consultancy, Honoraria; Imara: Consultancy, Honoraria; bluebird bio: Consultancy, Honoraria; Emmaus Life Sciences: Consultancy, Honoraria. Alvarez:Novartis: Membership on an entity's Board of Directors or advisory committees; Forma Therapeutics: Membership on an entity's Board of Directors or advisory committees. Lonshteyn:Policy Analysis Inc.: Current Employment; Global Blood Therapeutics: Research Funding; Novartis: Research Funding. Weycker:Policy Analysis Inc.: Current Employment, Current equity holder in publicly-traded company; Novartis: Research Funding; Global Blood Therapeutics: Research Funding. Pham:Policy Analysis Inc.: Current Employment; Global Blood Therapeutics: Research Funding; Novartis: Research Funding. Delea:Global Blood Therapeutics: Research Funding; Novartis: Research Funding; Policy Analysis Inc.: Current Employment, Current equity holder in private company. Agodoa:Global Blood Therapeutics: Current Employment, Current equity holder in publicly-traded company. Cong:Global Blood Therapeutics: Current Employment, Current equity holder in publicly-traded company. Shah:Novartis: Consultancy, Research Funding, Speakers Bureau; Global Blood Therapeutics: Consultancy, Research Funding, Speakers Bureau; CSL Behring: Consultancy; Alexion: Speakers Bureau; Bluebird Bio: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.